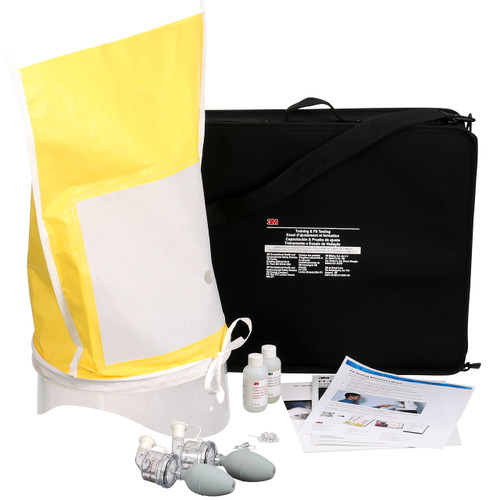
Description
The 3M 7100350838 FT-32 fit test solution is a ready-to-use bitter (Bitrex/denatonium benzoate) aerosol solution formulated for qualitative respirator fit testing. Supplied in compact 55 mL bottles, this 3M FT-32 solution is intended to be aerosolized through the qualitative fit test apparatus to verify facepiece-to-face seal under the OSHA respirator fit testing protocol. The formulation produces a detectable bitter taste for subjects who do not have an adequate seal, enabling clear pass/fail determination during user seal checks and fit-test procedures. Each bottle is packaged for occupational use and is commonly ordered in cases. The product is lightweight and easy to store, with handling and safety information provided on accompanying SDS documentation. Use the 3M FT-32 solution when you require a compact, single-use, or short-series fit-testing supply that integrates with 3M’s FT-30/FT-32 nebulizer systems and established qualitative fit-testing workflows.
Product Features and Benefits
Ready-To-Use Bitter Aerosol For Qualitative Testing
Pre-mixed Bitrex (denatonium benzoate) solution that requires no dilution—simply load into the nebulizer for immediate fit-testing, speeding test setup and reducing operator steps.
Compact 55 mL Bottle For Single Tests Or Small Batches
The 3M FT-32 fits small testing programs: a 55 mL bottle provides convenient handling, low shipping weight, and easy storage while supplying multiple fit tests depending on nebulizer use.
Designed For Use With 3M FT-30 / FT-32 Apparatus
3M FT-32 FIT Test Solution Its distinctive formula and packaging are matched to the 3M qualitative fit test apparatus; the solution aerosolizes properly in the FT-30/FT-32 nebulizers for consistent detection thresholds.
Meets OSHA Qualitative Fit Test Performance Criteria
Used within the standard qualitative fit test protocol to assess respirator facepiece seal in occupational settings, supporting compliance with 29 CFR
Case-Pack And Purchasing Details
Often supplied in case quantities, and minimum order thresholds vary by distributor; check distributor listing for pack quantity, SKU, and lead time before ordering.
Product Specifications
| Manufacturer Part Number | 7100350838 |
| Product Type | FT-32 Fit Test Solution |
Frequently Asked Questions
What volume is the 3M FT-32 fit test solution bottle?
Each FT-32 bottle contains 55 mL of solution.
Which nebulizer is 3M FT-32 compatible with?
The FT-32 solution is intended for use with 3M qualitative fit test apparatus such as the FT-30/FT-32 nebulizers.
Is this solution suitable for OSHA qualitative fit testing?
Yes, the FT-32 bitter solution is used for qualitative fit testing under OSHA 29 CFR 1910.134
What is the active agent used in the FT-32 solution?
The solution uses denatonium benzoate (Bitrex) as the bitter detection agent.
Warning:
This product can expose you to materials and/or chemicals including arsenic, lead, and other materials and/or chemicals which are known to the state of California to cause cancer and/or reproductive harm.
For more information, visit www.P65Warnings.ca.gov
 714.897.1700
714.897.1700
 Chat
Chat
 Email
Email
 Quotes
Quotes
 Quick Order
Quick Order